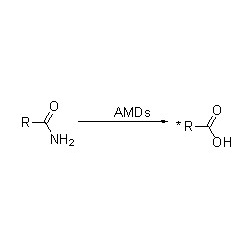
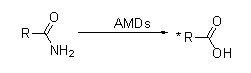
Amidase (AMD)
Enzymes: Are macromolecular biological catalysts, most enzymes are proteins
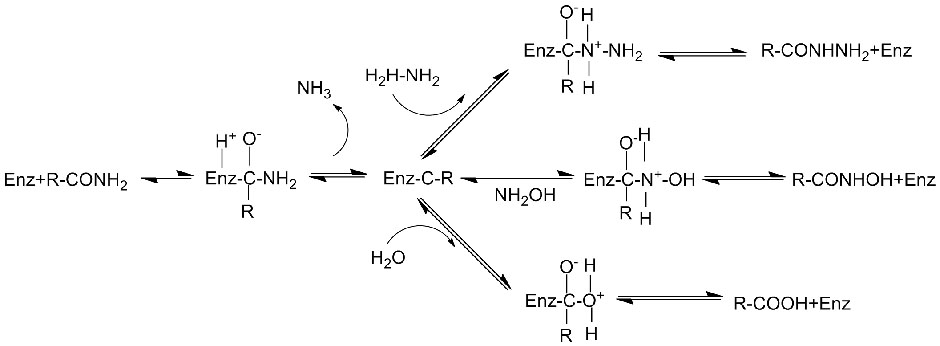
Amidase: Catalyze the hydrolysis of various endogenous and foreign aliphatic and aromatic amides by transferring an acyl group to water with the production of free acids and ammonia. Hydroxamic acids and other organic acids are widely used as drugs because they are constituents of growth factors, antibiotics and tumor inhibitors. The amidases can be divided into R type and S type acylases according to the catalyst stereoselectivity.
In addition to catalyzing the hydrolysis of amides, amidase can also catalyze acyl transfer reactions in the presence of co-substrates such as hydroxylamine.
Amidase with different sources have different substrate specificity, some of them can only hydrolyze aromatic amides, some of them can only hydrolyze aliphatic amides, and some hydrolyze α-or ω-amino amides. Most of the amines have good catalytic activity only for acyclic or simple aromatic amides, but for complex aromatics, heterocyclic amides, especially amides with ortho substituents, are generally low in activity (only a few enzymes exhibit better catalytic effects).
Catalytic mechanism:
| Enzymes | Product Code | Product Code |
| Enzyme Powder | ES-AMD-101~ ES-AMD-119 | a set of 19 amidases, 50 mg each 19 items * 50mg / item, or other quantity |
| Screening Kit (SynKit) | ES-AMD-1900 | a set of 19 amidases, 1 mg each 19 items * 1mg / item |
★ High substrate specificity.
★ Strong chiral selectivity.
★ High conversion efficiency.
★ Less by-products.
★ Mild reaction conditions.
★ Environmentally friendly.
➢ Enzyme screening should be carried out for specific substrates because of the substrate specificity, and get an enzyme that catalyzes the target substrate with best catalytic effect.
➢ Never contact with extreme conditions such as: high temperature, high/low pH and organic solvent with high concentration.
➢ Normally, the reaction system should include substrate, buffer solution (The optimum reaction pH of enzyme). Co-substrates such as hydroxylamine should be presence in acyl transfer reaction system.
➢ AMD should be added last into reaction system with optimum reaction pH and temperature.
➢ All kinds of AMD have various optimum reaction conditions, so each of them should be further studied individually.
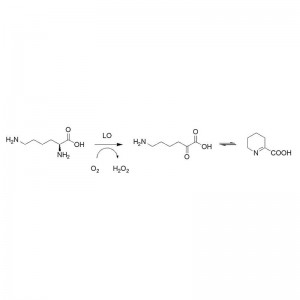
Example 1 (1):
Hydrolysis activity to different Amide Substrates
|
Substrate |
Specific activity μmols min-1mg-1 |
Substrate |
Specific activity μmols min-1mg-1 |
|
Acetamide |
3.8 |
ο-OH benzamide |
1.4 |
|
Propionamide |
3.9 |
p-OH benzamide |
1.2 |
|
Lactamide |
12.8 |
ο-NH2 benzamide |
1.0 |
|
Butyramide |
11.9 |
p-NH2 benzamide |
0.8 |
|
Isobutyramide |
26.2 |
ο-Toluamide |
0.3 |
|
Pentanamide |
22.0 |
p-Toluamide |
8.1 |
|
Hexanamide |
6.4 |
Nicotinamide |
1.7 |
|
Cyclohexanamide |
19.5 |
Isonicotinamide |
1.8 |
|
Acrylamide |
10.2 |
Picolinamide |
2.1 |
|
Metacrylamide |
3.5 |
3-Phenylpropionamide |
7.6 |
|
Prolinamide |
3.4 |
Indol-3-acetamide |
1.9 |
|
Benzamide |
6.8 |
The reaction was carried out in 50mM sodium phosphate buffer solution, pH 7.5, at 70 ℃.
|
Amides |
Hydroxylamine |
Hydrazine |
|
Acetamide |
8.4 |
1.4 |
|
Propionamide |
18.4 |
3.0 |
|
Isobutyramide |
25.0 |
22.7 |
|
Benzamide |
9.2 |
6.1 |
The reaction was carried out in 50mM sodium phosphate buffer solution, pH 7.5, at 70 ℃.
Related reagent concentration: amides, 100 mM(benzamide, 10 mM); hydroxylamine and hydrazine, 400 mM; enzyme 0.9 μM.
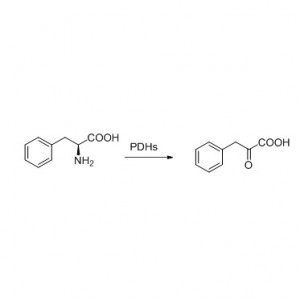
Example 2 (2):
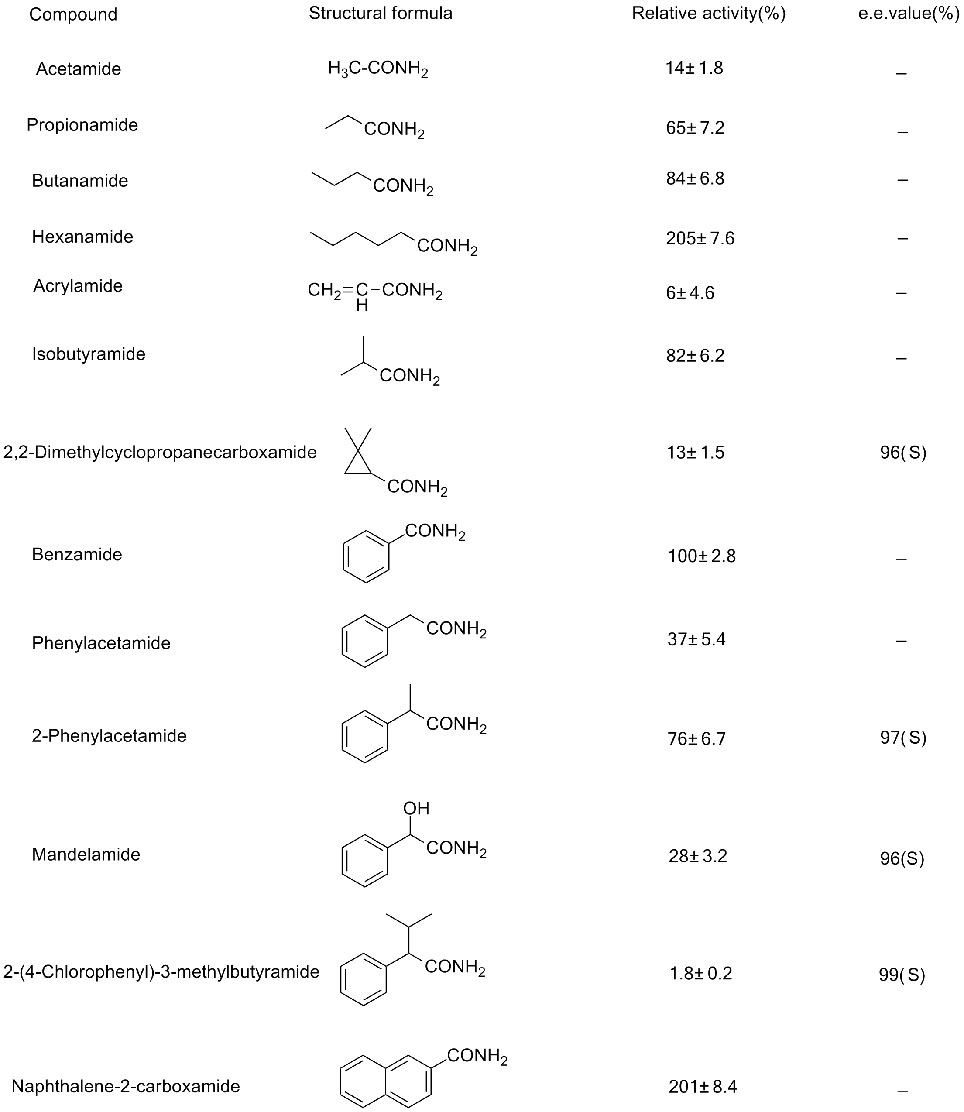
Example 3 (3):
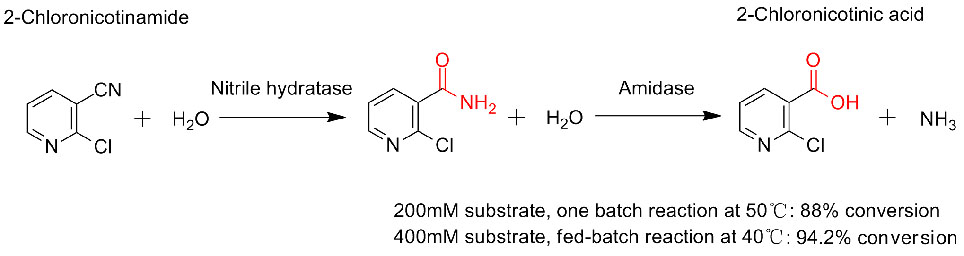
1. D'Abusco A.S., Ammendola S., et al. Extremophiles, 2001, 5:183-192.
2. Guo F M, Wu J P, Yang L R, et al. Process Biochemistry, 2015, 50(8): 1400-1404.
3. Zheng R C, Jin J Q, Wu Z M, et al. Bioorganic Chemistry, 2017, Available online 7.